First of all, let’s look at some of the glands and their effects. Naturally, the following explanations are not intended to be exhaustive.
Hypothalamus
The hypothalamus produces effector hormones, releasing and inhibiting hormones, various neuropeptides, and dopamine. It thus controls the vegetative functions of the body.
The hypothalamus is the most important control center of the vegetative nervous system which is comprised itself of a variety of homeostatic regulatory circuits. Even minute disruptions of this relatively small, but extremely important diencephalon (interbrain area) will have an effect on the viability of an individual. The functions of the entire vegetative system include the following:
Hypothalamus controls the vegetative nervous system.
- Maintaining homeostasis (temperature, blood pressure, osmolarity)
- Regulating the intake of food and water
- Circadian rhythms and sleep
- Control of sexual and procreational behavior (sex center)
Release of thyreoliberin (TRH)
TRH in turn stimulates the release of thyreotropin (TSH) which increases the body’s metabolic rate through the thyroid hormones.
Regulation of the body temperature:
Hypothalamus
-> TRH
->TSH
->Increase of metabolic rate through thyroid hormones
-> T4
-> T3
Ampoules:
Hypothalamus (Anatomy N/S 21) Hyperthyroidism (Harmful substances 171)
TRH (Hormones 1) Hypothyroidism (Harmful Substances 172)
TSH (Hormones 2)
T4 (Hormones 3)
T3 (Hormones 4)
Thyroid (Endocrine 8)
Thyroid stimulation (Endocrine 10)
Homeostasis of osmolarity
Vasopressin (Synonym: antidiuretic hormone, ADH, or adiuretin)
ADH increases the absorption of water from the primary urine. This in turn decreases the elimination of water by the kidneys, which counteracts hyperosmolarity and increases blood pressure.
Homeostasis of osmolarity
Hypophyseal portal system -> ADH -> Increased absorption of water from the primary urine
-> Decreased water elimination
-> Increased blood pressure
Ampoules:
Pituitary (Anatomy Endocrine/Urogenital 12)
Pituitary stimulation (Anatomy Endocrine/Urogenital 13)
ADH (Hormones 45)
Kidney R (Anatomy Endocrine/Urogenital 28)
Kidney L (Anatomy Endocrine/Urogenital 22)
[/fusion_text][fusion_text]Sleep and circadian rhythmsThe hypothalamus is also involved in regulating sleep and circadian rhythms.
The suprachiasmatic nucleus (SCN) receives direct afferents from the retina. Researchers assume that this is the seat of the “inner clock”, i.e. neurones that are responsible for the circadian rhythms. The SCN strongly controls the activity of the sympathicus. The SCN stimulates the release of melatonin from the pineal gland (epithalamus) via this vegetative system. Increased amounts of melatonin are released during the evening which contributes to sleep induction. The highest concentration in the blood can be found at three o’clock in the morning.
Sleep and circadian rhythms
Hypothalamus -> Suprachiasmatic nucleus -> Sympathicus
Pineal gland -> Melatonin
Ampoules:
Nervous exhaustion Harmful Substances I 2
Vegetative Dystonia Harmful Substances I 19
Insomnia Harmful Substances I 20
Depression melancholy Harmful Substances I 21
Melatonin Hormones 33
Vegetative NS Anatomy Nervous System/Senses 4
Hypothalamus Anatomy Nervous System/Senses 21
Epithalamus (pineal gland) Anatomy Nervous System/Senses 19
Influence on sexual behavior
The hormone oxytocin is released into the bloodstream. Oxytocin is involved in the contractions of the muscles of the womb during birth. It triggers the release of breast milk from the mammary glands and has a positive impact on partner bonding as well as mother and child bonding.[/fusion_text][one_fourth last=”no” spacing=”yes” center_content=”no” hide_on_mobile=”no” background_color=”” background_image=”” background_repeat=”no-repeat” background_position=”left top” border_size=”0px” border_color=”” border_style=”” padding=”” margin_top=”” margin_bottom=”” animation_type=”” animation_direction=”” animation_speed=”0.1″ class=”” id=””][fusion_text]
| Hormones of the hypothalamus |
| TRH (thyreotropin-releasing hormone, thyreoliberin) |
| CRH (corticotropin-releasing hormone, corticoliberin) |
| GnRH (gonadotropin-releasing hormone, gonadoliberin) |
| GHRH (growth hormone-releasing hormone, somatoliberin) |
| Dopamine (also described as a prolactin-inhibiting hormone) |
| Adiuretin (ADH, vasopressin) |
| Oxytocin |
| Effect on the pituitary gland |
| Release of TSH (thyroid-stimulating hormone, thyreotropin) |
| Release of ACTH (adrenocorticotropic hormone, adrenocorticotropin) |
| Release of FSH and LH |
| Release of growth hormone |
| Controls the release of prolactin |
| Posterior lobe of the pituitary gland |
| Posterior lobe of the pituitary gland |
| Endocrine gland / site of action |
| Thyroid |
| Adrenal cortex |
| Gonads |
| Mammary gland |
| Effect |
| Thyroxine and triiodthyronine |
| Aldosterone, cortisol, sex hormones |
| Inhibits lactation |
| Effector hormone: direct metabolic effect without gland |
| Effector hormone: direct metabolic effect without gland |
The pituitary gland (gr. hypóphysis „growth beneath“) is a hormonal gland which has a central superordinate role in regulating the neuroendocrine systems in the body. It is also called hypophysis.
We differentiate between hormones with a direct effect on the target organs (non-glandotropic hormones), and those that stimulate the production of hormones of endocrine glands downstream (glandotropic hormones).
- The growth hormone somatotropin (STH) as well as prolactin have a direct effect on the target organs.
- The glandotropic hormones are divided into gonadropic hormones, i.e. the follicle-stimulating hormone (FSH) and the luteinizing hormone (LH),
- and nongonadotropic hormones, i.e. the adrenocorticotropic hormone (ACTH), which stimulates the adrenal cortex and the thyroid-stimulating hormone (TSH), which stimulates the thyroid.
The hormones stored and released in the posterior lobe of the pituitary gland are oxytocin and the antidiuretic hormone (ADH), which is also known as adiuretin or vasopressin.
A diminished function of the pituitary (hypopituitarism, panhypopituitarism) can have several causes.
Tumors of the adenohypophysis are called pituitary adenomas. They are often the cause of excess hormone production.
Excess production of ACTH results in central Cushing’s disease, an excess of growth hormones which can usually be seen in enlarged hands and feet (acromegaly).
Epiphysis
The epiphysis produces the hormone melatonin. Hormone production takes places predominantly at night. Melatonin controls the sleep-wake rhythm and other time-dependent body rhythms.
Thyroid
The thyroid gland (lat. Glandula thyr(e)oidea) is located at the neck, underneath the voice box and in front of the windpipe. In humans it has a butterfly shape and lies underneath the thyroid cartilage in front of the windpipe.
The main function of the thyroid is the storage of iodine and the production of the iodine containing thyroid hormones thyroxine (tetraiodthyronine, T4) and triiodthyronine (T3) as well as the peptide hormone calcitonin.
Calcitonin inhibits bone loss by incorporating calcium and phosphate into the bone and by inhibiting the osteoclasts, which lead to a loss of bone substance if they are active.
Numerous diseases have their origin in the thyroid; among other things, they can be the cause of hormone metabolism problems as well as diminished or excessive function of the thyroid (hypothyroidism or hyperthyroidism). Iodine deficiency occurs frequently in the western world and can result in a goiter (struma) or lump.
The hormones triiodthyronine (T3) and thyroxine (T4) are very important for the proper development of a newborn’s organism. In adults, too, thyroid hormones influence the metabolism and functional status of nearly all organs.
The thyroid hormones are part of the so-called thyrotropic regulation circuit. Here, the function of the thyroid is regulated by the hypothalamus and the pituitary (anterior lobe of the pituitary gland). The hormone TSH (thyroid-stimulating hormone) is produced in the pituitary gland and released into the blood stream. When it reaches the thyroid cells it promotes their growth and the release of T3 and T4. T3 and T4 in turn inhibit the release of TSH. This mechanism is termed negative feedback and ensures that metabolic parameter are kept at a constant level in a healthy organism .
Thyroid hormones affect the heart and circulation. They increase the heart rate and blood pressure, and dilate vessels. They have an effect on the sugar, fat, and connective tissue metabolisms by increasing their transformation rate. They increase the activity of the skin’s sweat glands and sebaceous glands as well as intestinal activity. In the area of the nervous system, they result in an increased excitability of the cells. Overall the effect of the thyroid hormones increases energy consumption and the organism’s basic metabolic rate. This results in a rise in body temperature.
T3 and T4 communicate their effects via receptors in the target cells. Here, T3 is much more effective than T4. They thyroid cells predominantly produce T4, which is transformed (deiodinized) to T3 in the target cells. The receptors for thyroid hormones are mainly located in the cell nuclei and the mitochondria of the cells. They are proteins that are bound to the DNA of the genes they regulate and thus inhibit the gene expression. The receptors are activated by the binding of the thyroid hormones so that the gene expression of a whole range of proteins is facilitated or made possible in the first place.
Extensive thyroid therapy:
Cleansing of Toxoplasmosis
Candida
Fungus
Mycobacteria
Metals
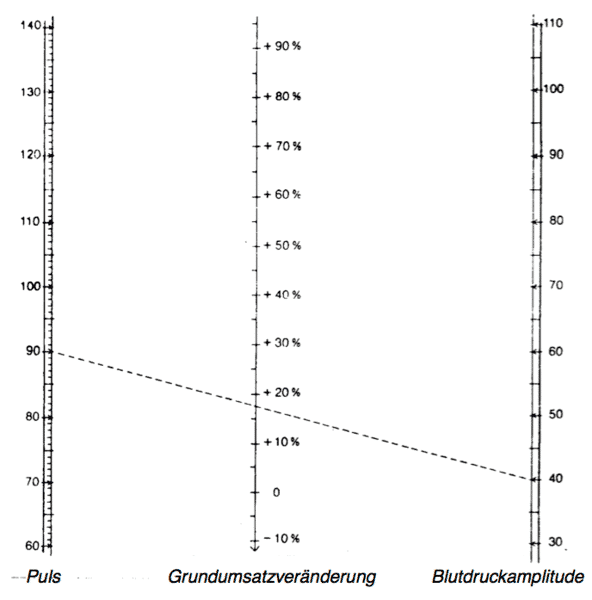
[/fusion_text][title size=”2″ content_align=”left” style_type=”default” sep_color=”” class=”” id=””]Nomogram for Read’s formula[/title][fusion_text]Calculating the increase in basal metabolic rate from pulse rate and blood pressure amplitudeIn cases of hyperthyroidism (Basedow’s disease) there is a certain regular relationship between heart frequency and the size of the blood pressure amplitude, which is the basis of Read’s Formula, which has been proven time and again when it comes to an approximate determination of the basal metabolic rate.
Read’s Formula: BMR = 0.75 (p+0.74a)-72
With:
BMR = basal metabolic rate in percent (of the norm)
p = pulse rate
a = blood pressure amplitude = difference between maximum and minimum pressure
The nomogram depicted here saves having to execute the calculation that would result from applying Read’s Formula. If one joins the point on the left scale, which corresponds to the determined pulse rate, with the point on the right scale, which is determined by the measured blood pressure amplitude, the point of intersection of this connective line with the middle scale shows the increase in percentage of the basal metabolic rate compared to the norm – as calculated by the application of Read’s Formula.[/fusion_text][/one_full][fusion_text]If excess -> Secondary hyperthyroidism
Pituitary gland produces TSH -> Growth, iodine uptake, hormone production by thyroid
If deficiency -> Secondary thyroid function (hypothyroidism)
Parathyroid
The parathyroid, two organ pairs, produce the parathyroid hormone, a hormone that increases the calcium level in the blood.
The main cells produce the parathyroid hormone (PTH). The parathyroid hormone, as a calcitonin antagonist, increases the calcium concentration in the blood by indirect activation of osteoclasts.
Hypofunction of the parathyroid is less frequent (hypoparathyroidism); these are usually iatrogenic, e.g. after thyroid surgery or oversupply with vitamin D. Autoimmune diseases can also trigger hypothyroidism. One symptom is a parathyroid hormone deficiency that can lead to hypocalcemia with cramps and heart failure.
Stomach
Hormone-producing endocrine cells as well as exocrine glandular cells, which produce mucous, are integrated in the epithelium of the pylorus glands:
The hormone gastrin is produced here, which stimulates acid production in the parietal cells. This happens directly and indirectly through stimulating the release of histamine.
The D-cells produce somatostatin. They are stimulated by stomach acid in the stomach lumen. Somatostatin inhibits the release of gastrin and histamine as well as directly also the acid production in parietal cells.
Ampoules : Gastrin Hormones 29
Histamine Interior Milieu 24
Stomach entry Filter Organ Human 2
Stomach floor Filter Organ Human 3
Phylorus (stomach exit) Filter Organ Human 4
Stomach extension Harmful Substances 136
Stomach vomiting Harmful Substances 137
Stomach cramps Harmful Substances 138
Gastritis Harmful Substances 140
Stomach ulcers Harmful Substances 141
/Ulcus ventriculi
Gastroduodenitis Pathologies 3
Ulcus duodeni Pathologies 4
Ulcus ventriculi Pathologies 5
Pancreas
The pancreas is a glandular organ lying horizontally in the upper part of the abdomen. The digestive enzymes produced by it are released into the duodenum through one or two excretory ducts. It is therefore an exocrine gland (exocrine “secreting outward”; in this case, into the digestive tract).
These digestive enzymes split the proteins, carbohydrates, and fats in food into their basic components inside the intestines and thus reduce them to a size that is absorbable by the intestinal mucosa.
In addition, there are hormones produced in the pancreas that are secreted directly into the blood. This means that it is also an endocrine gland (endocrine “secreting inward”). This endocrine part of the pancreas is called the islets of Langerhans, which are primarily responsible for regulating the blood sugar levels (through the hormones insulin and glucagon) as well as digestive processes.
An inflammation of the pancreas (pancreatitis) leads to self-digestion through the released digestive enzymes. If the exocrine part stops functioning properly (exocrine pancreatic insufficiency), food can no longer be broken down properly. The most well-known disorder of the endocrine part is diabetes (diabetes mellitus).
Ampoules : Ductus pancreaticus GI Tract/ Immune System 10
Pancreas exocrine Pathologies 26
Pancreas endocrine Pathologies 27
Pancreas Filter Organ Human 8
Pancreatitis. Harmful Substances 150
Pancreas disorder Harmful Substances 151
Blood sugar regulation Harmful Substances 152
Sugar diabetes mellitus Harmful Substances 153
Insulin Hormones 30
Glucagon Hormones 31
Diabetes pathogen:
Coxsackie viruses + Viruses general + Candida
(Viruses/Borrelia 29-39) (Mycoses1,3,4,5,7,9,10)
Eurythrema pancreaticum + Other leeches + Fungi
Parasites 18 Parasites 1-23
Heavy metals
[/fusion_text][fusion_text]KidneyThe kidney also produces hormones: renin (enzyme, short-term blood pressure regulation), erythropoetin (stimulates blood formation), calcitriol (vitamin D, plays a role in the calcium metabolism), kinins, and prostaglandins.
Adrenal gland
The adrenal gland (Latin: Glandula adrenalis or Glandula suprarenalis) is a paired hormonal gland. In humans the adrenal glands are found above both kidneys. They are subject to the hormonal regulatory cycle and the vegetative nervous system.
The adrenals functionally combine two different organs:
- The adrenal cortex produces steroid hormones and plays a role in regulating water, mineral, and blood sugar levels.
- The adrenal medulla is part of the sympathetic nervous system and produces adrenaline and noradrenaline.
All adrenal cortex hormones are synthesized from cholesterol. The cholesterol is transported to the mitochondrial inner membrane. There it is transformed into pregnenolone. Pregnenolone can either be dehydrated to progesterone or hydroxylated to 17-alpha-hydroxypregnenolone. Through hydroxyation progesterone can be converted to deoxycorticosterone and through further hydroxyations to aldosterone. Progesterone can be hydroxylated to 17-alpha-hydroxyprogesterone and then from deoxycortisol to cortisol.
The adrenal medulla (Latin: Medulla glandulae suprarenalis) uses L-tyrosin to produce adrenaline (about 80 %) as well as noradrenaline (about 20 %) and secretes this directly into the blood if required.
Liver
The liver is closely involved in regulating the glucose, fat, and protein metabolisms. Glucose is absorbed by the intestinal blood and distributed to the rest of the body in a controlled manner. Any excess is stored as glycogen. The stores are converted to glucose when hungry. Controlled by hormones like insulin and glucagon, the liver influences blood sugar levels and can keep them constant, independent of any food intake. Insulin effects the conversion of sugar to the storage form glycogen in the liver and inhibits the catabolism of fat. The hormone glucagon in turn stimulates the liver to break down glycogen and thus acts as a counterpart to insulin.
The prohormone angiotensin I (a link in the renin-angiotensin-aldosterone system responsible for maintaining blood pressure and water balance) is produced in the liver tissue by renin enzymatically from angiotensinogens.
Gonads
The two ovaries are primary female sex organs. As a gonad it corresponds to the testicles in male individuals and is the place where the eggs (ova) and female sex hormones are produced.
The testicles are the inner male sex organs of many sexually reproducing eumetazoa. Just like the ovaries in female individuals, it belongs to the so-called gonads. It produces the seminal filaments (sperm). Additionally, male sex hormones (androgens) are produced in the testicles, especially testosterone.
[/fusion_text][title size=”3″ content_align=”left” style_type=”default” sep_color=”” class=”” id=””]Endocrine cascades: hypothalamic-pituitary axis:[/title][fusion_text]Prolactin (milk production of breast)Hypothalamus To anterior pituitary
Produces thyroliberin -> (Anterior lobe pituitary gland)
(TRH) TSH
Thyroid-stimulating hormone
Is stimulated by temperature
(L-thyroxine) (Triiodothyronine)[/fusion_text][fusion_text]
- The hypothalamic-pituitary-thyrotroph axis:
Thyroliberin, also called thyreotropin-releasing hormone (TRH), is produced by the hypothalamus. It stimulates the production and release of prolactin and TSH. Thyroliberin (via TSH) therefore also stimulates the release of the thyroid hormones T4 and T3 in the thyroid.
A lowering of the body temperature, among other things, stimulates the release of TRH: The metabolism is stimulated by the subsequent TSH and the then following T4 release, which causes the body temperature to rise again through an increased sugar metabolism. Other energy-demanding mechanisms also stimulate the release of TRH.
The TRH release happens in a circadian rhythm, with a maximum release at around midnight and a minimum release late in the afternoon. The rhythmic release of TRH is furthermore influenced by the limbic system, the pineal gland, and other brain regions that are important for the stress response.
Apart from its main effect on the pituitary gland, thyroliberin also stimulates the release of prolactin.
As a neurotransmitter in the brain, thyroliberin is involved
- in thermoregulation,
- suppressing of pain,
- sleep-wake regulation,
- slowing down the uptake of food and liquid, and
- a myriad of other control processes.
Thyreoliberin also has indirect vegetative effects:
- It stimulates the production of stomach acid and intestinal peristalsis via the vagus nerve.
- It stimulates insulin secretion via the sympathicus.
- It also stimulates the pancreas and its exocrine functions.
- It increases heart rate and blood pressure.
L-thyroxine T4 is necessary to treat an underactive thyroid (hypothyroidism). People suffering from this disease usually require life-long hormone substitution.
Thyroxine and triiodothyronine moreover play a significant role in effect amplification and mood stabilizing when treating depression.
Triiodothyronine is one of two very important thyroid hormones. It is the most effective thyroid hormone and has an effect 3-5 times greater than the second iodine-containing thyroid hormone tetraiodothyronine (T4).
Calcitonin is the counterpart to the parathyroid hormone, which is produced in the parathyroid. Both hormones regulate the calcium and phosphate balance in the body. Additionally, calcitonin has a calcium-lowering effect.
Calcitonin lowers the blood calcium levels: It inhibits the release of calcium from the bone (by decreasing osteoclast activity), promotes calcium excretion via the kidneys, and lowers intestinal calcium absorption. However, renal phosphate excretion also raises it, as does the parathyroid hormone.
Furthermore, it acts as an analgesic in bone metastases. Calcitonin is used as a tumor marker in medullary thyroid cancer.
The parathyroid hormone (PTH) / parathyrin is a peptide hormone consisting of 84 amino acids. The main function of the parathyroid hormone is to increase the concentration of calcium in the blood plasma.
Indirectly PTH leads to a maturing and activating of the osteoclasts and thus to a calcium phosphate mobilization from bone issue. PTH inhibits the phosphate absorption in the kidneys.
The blood phosphate level also decreases because more phosphate is excreted with the urine. This makes sense, because it means that more free ionized calcium is available again in the blood (which, if the phosphate concentration is too high, forms a complex in the kidneys which is difficult to dissolve; it subsequently breaks down and leads to a calcification of the kidneys).
Increased blood levels (hyperparathyroidism):
- Irregular epithelial corpuscles (adenomas or, rarely, carcinomas) are no longer subject to calcium-dependent secretion control. The result is an uncontrolled rise in parathyroid hormone levels (primary hyperparathyroidism) and subsequently hypercalcemia.
- The body reacts to hypercalcemia because of a disease of the kidney, liver, or intestines with an increased secretion of parathyroid hormones (secondary hyperparathyroidism).
- Within the context of malignant diseases (bronchial carcinoma, breast carcinoma, prostate carcinoma, plasmacytoma) paraneoplastic hypercalcemia can occur. The irregular cells produce a peptide (PTHrP) closely related to the parathyroid hormone that has a similar effect to it (pseudohyperparathyroidism).
Decreased blood levels (hypoparathyroidism):
- Thyroid surgery, removal of epithelial corpuscle adenoma, or an autoimmune disease can result in parathyroid hormone insufficiency. The calcium levels drop and hypocalcemic tetany ensues.
Also as Calcitonin D parathyroid hormone
Tumor marker are antagonists
Thyroid gland Parathyroid gland Inclusion of Increased calcium level
Calcium and phosphate
in bone Break down in bones ![/fusion_text][fusion_text]
- The hypothalamic-pituitary-adrenal axis:
The corticotropin-releasing hormone (CRH) is produced in the hypothalamus. This in turn stimulates, among other things, the release of ACTH (adrenocorticotropic hormone) and the sympathicus.
The release of CRH is subject to a circadian rhythm (increased secretion in the morning, less in the evening), as well as impulses from the limbic system and the negative feedback by the glucocorticoids produced under the influence of ACTH.
CRH also seems to be involved with many other central and peripheral processes. For example, it is assumed that it plays a part in the following systems and processes: cardiovascular, inflammation, reproduction, pregnancy, thermoregulation, nutritional intake, and the psyche.
ACTH is produced under the influence of corticotropin-releasing hormones (CRH) in the anterior lobe of the pituitary gland. ACTH is a melanocortin receptor agonist in the adrenal cortex. There, it stimulates the adrenal cortex to produce glucocorticoids, mineralocorticoids, and sex hormones.
Raised ACTH levels can be the result of cold, stress, adrenal cortex insufficiency, Cushing’s disease, or paraneoplastic syndrome. Reduced ACTH levels occur for Sheehan’s syndrome, changes to the pituitary, hypothalamus, or pituitary stalk. Possible changes to the pituitary can be the result of pituitary or brain tumors, surgery, radiation, bleeding, infarctions, infections, inflammations, granuloma, or metastases in the area of the pituitary. Possible changes to the hypothalamus, which regulates ACTH production in the pituitary with the help of CRH, are due to tumors, surgery, or radiation. If the pituitary stalk is injured, ACTH levels will decrease since the hypothalamus can no longer drive the production of ACTH in the anterior lobe of the pituitary gland using CRH.
Tears from crying (for emotional reasons, not because of irritation to the eyes) contain adrenocorticotropin.
ACTH is also called a stress hormone, because different forms of stress can increase the release of it. Possible stressors are work, injuries, sickness, surgery, emotions, depression, physical and psychological stresses.[/fusion_text][fusion_text]Hypothalamic-pituitary-adrenal axis
|
Hypothalamus
Stressors – stimulation
CRH Stimulates ACTH
Sympathicus Adrenal cortex
|
|
Increased levels for:
Negative feedback[/fusion_text][fusion_text]Glucocorticoids, hormones from the adrenal cortex. Naturally occurring glucocorticoids are derivatives of progesterone. Among these are cortisol and corticosterone, with a percentage of 95 % and 5 %, respectively, in humans.
Glucocorticoids have manifold physiological effects. They influence the metabolism, fluid and electrolyte balance, cardiovascular system, and the nervous system. Moreover, they are anti-inflammatory and immunosuppressive.
The biosynthesis of corticoids starts, as for all steroids, with cholesterol, which is either taken up through your food or is formed (in much larger amounts) from mevalonate. Pregnenolone is the intermediate, which is converted in several possible ways to cortisol, and from this to cortisone. There is a distinct day rhythm with a minimum around midnight and a maximum between 6 and 8 o’clock in the morning.
Like other corticoids, glucocorticoids are inactivated in the liver and mainly excreted through bile fluid, with up to 10 % also through urine.
Glucocorticoids in natural concentration promote gluconeogenesis, i.e. the generation of carbohydrates from proteins. Protein and lipid depots are broken down and used to generate energy. This is shown in increased concentrations of glucose, amino acids, and fatty acids in the blood, as well as their metabolic products. Chronic overproduction of these hormones leads to a disease known as Cushing’s syndrome.
Cortisol is a hormone that activates catabolic metabolic processes and thus makes energy-rich compounds available to the body. Its dampening effect on the immune system is often utilized in medicine to suppress exaggerated reactions and inhibit inflammations.
The production in the adrenal cortex is stimulated by the adrenocorticotropic hormone (ACTH). A hyperfunction leads to the clinical picture of Cushing’s syndrome; a diminished function, or hypofunction, is called Addison’s disease.
Cortisol has a very broad action spectrum. Metabolically speaking, its main effects are on the carbohydrate balance (promoting gluconeogenesis in the liver), fat metabolism (promoting the lipolytic effect of adrenaline and noradrenaline), and protein turnover (catabolic). Cortisol has a similar effect to aldosterone and is thus oxidized to cortisone in the kidneys, intestines, and a few other tissues. Cortisone does not bind to a mineralocorticoid receptor and has therefore no antidiuretic effect. This means it does not prevent the excretion of poisonous substances through the urine. If there is a lack of functioning adrenal cortex tissue, cortisol must be substituted.
Higher instances of cortisol secretion take place in the hypothalamus via the pituitary gland. Here, the hypothalamus releases CRH which results in the release of ACTH (adrenocorticotropic hormone) in the anterior pituitary (anterior lobe of the pituitary gland). The remarkable thing with these hormones is their pulsatile release, i.e. they are secreted in regular batches (7-10 per day). Cortisol levels show typical fluctuation in the course of a day (circadian rhythm). The highest level is reached in the morning, shortly after waking up (cortisol awakening response, CAR).
In higher doses, cortisol is anti-inflammatory and immunosuppressive.[/fusion_text][fusion_text]Hypothalamus
Pituitary gland
ACTH Hyperactivity Cushing’s disease
Cortisol (NNR)
Underactive Addison’s disease
Gluconeogenesis in the liver (carbohydrate balance)
Fat metabolism… Promoting the lipolytic effect of adrenaline and noradrenaline
Protein rate (catabolic)[/fusion_text][fusion_text]Mineralocorticoids are part of the corticosteroids, a class of adrenocortical steroid hormones. The two most important mineralocorticoids are aldosterone and desoxycorticosterone. Just like the glucocorticoids, mineralocorticoids are derivatives of progesterone.
Mineralocorticoids are similar to glucocorticoids, but they predominantly influence the fluid and mineral balance in the body.
Mineralocorticoids increase the reabsorption of sodium in the kidneys and increases the excretion of potassium. Water excretion follows sodium, therefore mineralocorticoids increase the water levels in the body. The biosynthesis of the corticoids begins, as with all steroids, with cholesterol. Pregnenolone is the intermediate which is converted to corticosterone and aldosterone. Like other corticoids, mineralocorticoids are inactivated in the liver and mainly excreted through bile fluid, and up to 10% also through the urine.[/fusion_text][fusion_text]Mineralocorticoids aldosterone Mineral balance
(Derivative Desoxycorticosterone Water balance
of progesterone)
Sodium reabsorption in kidneys # Water content #
Potassium excretion through kidneys #
Promote gluconeogenesis Generation of carbohydrates from protein
Protein and lipid depots are broken down
Energy generation Glucose #
Amino acids #
Fatty acids #
|
Glucocorticoids
Cortisol + corticosterone
Inactivated in the liver
Excreted through bile
10% excreted through urine[/fusion_text][fusion_text]Aldosterone is a natural steroid hormone produced from cholesterol and is part of the mineralocorticoids. Aldosterone is sometimes called the thirst hormone, because of its increased secretion if there is a lack of fluids.
Aldosterone is produced in the adrenal cortex. The release of aldosterone increases when blood volume and blood pressure decrease (conveyed through angiotensin II via AT1 receptors), and hyperkalemia. The sodium concentration in the blood has an opposing influence on aldosterone synthesis: if blood sodium levels rise, biosynthesis is inhibited; if sodium falls, it is stimulated. ACTH stimulates aldosterone synthesis while atriopeptin (ANP) inhibits the release.
Overall aldosterone effects an increase in extracellular volume, a decrease in potassium concentration, and a rise in the blood pH-value.
One important role played by aldosterone and the renin-angiotensin-aldosterone system is that of blood pressure regulation. Furthermore, aldosterone seems to be necessary in coping with life-endangering stress situations.
Short-term effects of aldosterone that occur within minutes or hours serve to maintain stability of fluid, electrolyte, and acid-base balances (interior milieu) by increasing the reabsorption of sodium as well as excretion of potassium and protons (physiological effects).
However, chronically high aldosterone levels – for days, weeks, or months – have unfavorable effects. The result is a potassium deficiency (hypokalemia), a rise in pH-value (alkalosis), high blood pressure, and the increased formation of tissue (fibrosis) in the heart and kidneys; this can eventually lead to chronic heart and kidney insufficiency (pathophysiologic effects).
Aldosterone levels are low for adrenal cortex insufficiency and shock, while aldosterone levels are increased during stress, after surgery, and for increased angiotensin II activity (hyperaldosteronism).
Primary hyperaldosteronism (Conn’s syndrome) is rare; however, secondary hyperaldosteronism can be found frequently in the case of heart insufficiency and cirrhosis of the liver. Unfavorable effects of aldosterone were also described for acute heart infarction, nephrotic syndrome, intractable hight blood pressure, chronic kidney failure, in particular for diabetic nephropathy, and metabolic syndrome.
A lack of aldosterone might occur, for example, as part of an autoimmune disease (Addison’s disease) or in the case of a tuberculosis of the adrenal glands.
Aldosterone (mineralocorticoid) Thirst hormone in the event of lack of fluids
Adrenal cortex
Decrease in blood volume
Of blood pressure Aldosterone # <=> Sodium concentration in the blood #
Hyperkalemia (inhibits)
pH level in blood#
Angiotensin I is a link in the renin-angiotensin-aldosterone system (RAAS) which is responsible for maintaining blood pressure and water balance. Angiotensin is a mixture of inactive angiotensin I and the vessel-constricting angiotensin II.
Angiotensin II is a tissue hormone made up of eight amino acids. It has a key position in the renin-angiotensin-aldosterone system (RAAS), which is responsible for maintaining blood pressure and water balance.[/fusion_text][/fullwidth]